Program Goals
Identify and contain AR organisms
Promote appropriate use of antimicrobials
Submit data submission to health care facilities for targeted intervention to decrease HAIs
- Promote sustainable prevention infrastructure in health care facilities across the state
Objectives
Identify and contain emerging pathogens and antibiotic resistant organisms of interest
Improve outbreak identification, investigation and reporting
Use the Center for Disease Control (CDC) National Healthcare Safety Network for surveillance of HAIs
Support implementation of antimicrobial stewardship in health care facilities
Provide education, mentorship and support to infection preventionist in all health care settings
- Facilitate evidence-based practices for infection prevention and control in health care settings through education and training, consultation and access to resource materials.
Health Alert Network (HAN) Alert-Candida auris
On May 13, 2021, a HAN communication was issued in Kentucky. An updated HAN was issued on July 15, 2021. HAN Alert for C. auris
Required Reporting-Updated
HAIs and Multidrug Resistant Organisms (MDROs) reporting regulation -
902 KAR 2:020 Reportable disease surveillance
Changes in reporting requirements for all MDROs have been made.
- Reporting within one business day of:
- Candida auris with
- Confirmatory lab evidence (PCR or culture)
- Form EPID 250 (fax 502-398-2462)
Reporting within one business day with EPID 250 (fax (502) 398-2462) and labs with Minimum Inhibitory Concentration (MIC) values of:
- Carbapenem-resistant
- Acinetobacter
- Enterobacteriales
(Enterobacteriaceae)
- Pseudomonas
- Vancomycin-intermediate Staphylococcus aureus (VISA)
- Vancomycin-resistant Staphylococcus aureus (VRSA)
- Electronic lab reporting within five days is required for (no form EPID 250 required):
- Clostridioides difficile (C. difficile)
- Enterobacteriales (Enterobacteriaceae) species resistant to ceftazidime, ceftriaxone or cefotaxime (ESBL)
- Methicillin-resistant Staphylococcus aureus (MRSA)
- Vancomycin-resistant Enterococcus species (VRE)
Kentucky Infection Prevention (KyIP) Training Center
The KyIP Training Center provides education and training to all health care professionals including nurses, infection preventionists, physicians, pharmacists, and allied health professionals, along with anyone interested in the areas of infection prevention and control across the Commonwealth of Kentucky. A link to their webpage can be found here.
What is an HAI?
HAIs are infections patients develop while receiving healthcare. HAIs can occur in all health settings including, but not limited to, hospitals, ambulatory surgical centers, dialysis facilities, outpatient clinics e.g., physicians' offices, health care clinics , rehabilitation centers and nursing homes. HAIs can be associated with procedures and the devices used in medical care and treatment.
Common HAIs include: central line associated bloodstream infection, catheter-associated urinary tract infection, surgical site infection, Clostridioides difficile and methicillin-resistant Staphylococcus aureus infections.
What is Antimicrobial Resistance?
Antimicrobials are powerful tools for fighting and
preventing infections. However, widespread use of antimicrobials has resulted
in an alarming increase in antimicrobial-resistant infections. Resistance
occurs when microorganisms, including bacteria, viruses, fungi, and parasites
evolve in a way that reduces the effectiveness of drugs designed to cure or
prevent infections. As antimicrobial-resistant infections become more common,
therapeutic options for the treatment of infections are increasingly limited,
expensive, and often more toxic. Antimicrobial stewardship is a critical
strategy for combating the growing problem of antimicrobial resistance by
optimizing the use of antimicrobials and safeguarding patient safety and
healthcare outcomes. For more information, please visit our page on antimicrobial
resistance and stewardship.
Regional Infection Preventionists
To better assist in the response to the COVID-19 pandemic, the department recently added regional infection preventionists. These regional IPs work with facilities in the local communities to help respond to and control the spread of COVID-19 in the health care setting.
Kentucky is divided into 10 regions, each with a regional infection preventionist. See map and table below for contact information.
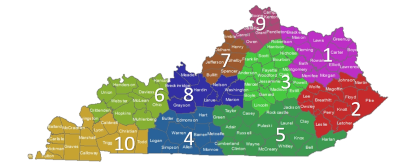
Kentucky State-Regional Infection Prevention and Epidemiology
Kentucky-State Regional Infection Prevention and Epidemiology advises the HAI/AR Prevention Program. Kentucky Hospital Association is a long-term partner of the HAI/AR Prevention Program, working on issues of infection prevention and control in acute care and critical access hospitals and providing education to infection preventionists in Kentucky.
More information about the program can be found in the
State HAI Plan .
Multi-drug resistant organisms (MDRO)
MDROs are common germs, usually bacteria, resistant to multiple antibiotics. MDROs can cause active infections with or without symptoms.
MDROs commonly spread by direct contact between people or with contaminated surfaces in the environment. MDROs can be difficult to treat, depending on the antibiotics to which the bacteria are resistant.
MDRO Reporting
MDRO reporting has been required since 2016 and involves completing the multidrug-resistant reporting form EPID 250 (fillable form:
EPID 250 Fillable) and providing the associated laboratory results.
Instructions for filling out the form EPID 250 are provided, as are more in-depth
definitions for carbapenem-resistant organisms.
The MDRO Reporting mandate has been updated. Please see updated version above.
New Recognized Infectious Agents, HAI Outbreaks, Emerging Pathogens and Pathogens of Public Health Importance
An outbreak of a disease or condition that resulted in multiple hospitalizations or death is expected to be reported immediately by phone to the Kentucky Department for Public Health.
An unexpected pattern of cases, suspected cases or deaths which may indicate a newly-recognized infectious agent; an outbreak or epidemic; an emerging pathogen posing a public health danger; or a non-infectious chemical, biological or radiological agent. These events are to be reported to the Kentucky Department for Public Health by phone.
Outbreaks and Investigations
As reports are processed the HAI/AR Prevention Program will conduct outbreak investigations when indicated. An HAI outbreak is defined as: the occurrence of two or more HAIs or MDROs epidemiologically linked or connected by person, place or time; or a single case of an HAI not commonly diagnosed. An investigation also may occur if a specific HAI or MDRO is found in a location for the first time.
Response
Investigations are traditionally focused on outbreaks of MDROs and emerging pathogens. Additionally, there have been investigations related to infections occurring related to devices e.g., intrathecal pain pumps and outpatient procedures intra-articular joint injections. Site visits are conducted when possible. We engage CDC when emerging pathogens/mechanisms are identified. We use the Kentucky Division of Laboratory Services and CDC lab for pulsed field gel electrophoresis and whole-genome sequencing when needed.
Point Prevalence Survey (PPS)
A PPS involves screening at-risk individuals when there is concern for potential horizontal transmission of MDROs between patients either directly or via healthcare providers. The Antibiotic Resistance Laboratory Network provides assistance when PPS are performed. The AR Lab Network national initiative is funded through CDC grants.
If within-facility transmission is identified or suspected, PPS will continue until transmission is halted. Additionally, a targeted onsite assessment will be performed if possible and assistance provided to the facility to mitigate breaches of infection prevention and control. Re-emphasis on facility infection prevention and control activities always is part of the response.
Target Infections Control Assessment and Response
Infection Control Assessment Tools were developed by CDC under the Epidemiology and Laboratory Capacity Infection Control Assessment and Response Program to help health departments assess infection prevention practices and guide quality improvement activities, such as addressing identified gaps. These tools also may be used by health care facilities to conduct internal quality improvement audits.
Center for Disease Control National Healthcare Safety Network (NHSN)
What is the CDC NHSN? It is a national HAI tracking system that provides data to identify problem areas, measure progress of prevention efforts and ultimately eliminate health care-associated infections.
The network allows health care facilities to track HAIs as well as blood safety errors and important health care process measures such as healthcare personnel influenza vaccine status and infection control adherence rates.
Target, Assess and Prevent (TAP)
The
Targeted Assessment for Prevention (TAP) Strategy is a framework for quality improvement developed by the CDC to use data for action to prevent HAIs. The TAP Strategy consists of three components: 1. running TAP reports in the National Healthcare Safety Network (NHSN) to target healthcare facilities and specific units with an excess burden of HAIs; 2. administering TAP facility assessment tools to identify gaps in infection prevention in the targeted locations; and 3. accessing infection prevention resources in TAP implementation guides to address those gaps. The TAP assessment survey is administered to a variety of people in the hospital to identify breaches in knowledge for specific infection control practices and activities.
Extensively Drug Resistant Organism Registry (XDRO)
The XDRO registry is populated with patient information and laboratory data for all carbapenem-resistant Enterobacteriaceae CRE identified in Kentucky facilities or identified in Kentucky residents. The registry can be queried by facility infection preventionists to support appropriate transmission-based precautions when patients have healthcare contact. Initial entry in the XDRO registry is made manually by the HAI/AR Prevention Program staff, although eventually the registry will be populated automatically from electronic laboratory reporting through the Kentucky Health Information Exchange (KHIE) . Additionally, an alert feature is planned to allow real-time use of admission, transfer and discharge data in KHIE to alert facilities of patient CRE history. This will provide more timely initiation of appropriate transmission-based precautions and limit opportunities for spread of the organism in the facility.
If you are interested in gaining access to the database please contact
Brittany Corley.
Antimicrobial Stewardship
According to Society for Healthcare Epidemiology of America (SHEA) , antibiotic stewardship involves coordinated strategies to improve use of antimicrobial medications to enhance patient health outcomes, reduce resistance to antibiotics and decrease unnecessary costs.
Antimicrobial stewardship includes a systematic effort to educate and persuade prescribers of antimicrobials to follow evidence-based prescribing.
Statewide antimicrobial stewardship report 2013-14
Collaborative Activities
- The Louisville-Metropolitan Antimicrobial Resistance/Antibiotic Stewardship Collaborative serves as a vehicle for discussion of issues related to infection prevention and control, antimicrobial resistance and antibiotic stewardship. Participants include physicians, infection preventionists, microbiologists and others from acute and long-term care facilities in the Louisville metropolitan area. Goals include strengthening inter-facility communication and improving consistency of response to antimicrobial resistance threats.
- Southeastern Kentucky Regional Collaborative brings together healthcare facilities in the region to discuss issues of infection prevention, antibiotic resistance and interfacility communication
- Work with Alliant Quality is a quality-improvement group for Alliant Health Solutions.
- The hospital-onset (HO) Methicillin-resistant Staphylococcus aureus (MRSA) Bloodstream Infection Collaborative with CDC and Tennessee is an effort to reduce rates of MRSA bacteremia in facilities with historically higher rates through targeted evidence-based interventions.
- Kentucky Hospital Association is a long-term partner of the HAI/AR Prevention Program, working on issues of infection prevention and control in acute care and critical access hospitals and providing education to infection preventionists in Kentucky.
- Work with the Child and Adolescent Health Research Design and Support Unit (CAHRDS) Antibiotic Team, Department of Pediatrics, University of Louisville School of Medicine. This group analyzes Medicaid prescribing and claims data related to antibiotic prescribing in pediatrics to improve outpatient antibiotic prescribing for pediatric patients across the state. More generally, it actively is involved in increasing public and provider knowledge about antibiotic use and antimicrobial resistance as critical public health issues.
Frequently Asked Questions
What is the difference between an active infection and colonization with an organism?
Active infection indicates an organism (bacteria, virus, etc.) is found in or on the body and is associated with signs and/or symptoms of illness. Colonization indicates that an organism is found in or on the body but is not producing signs or symptoms of illness.
Are contact precautions necessary in long-term care?
According to the
CDC place CRE colonized or infected residents that are high-risk for transmission on CP. For patients at lower risk for transmission use precautions based on type of care provided.
Resources